Detection of Mycobacterium tuberculosis complex and Rifampin- resistance associated mutations in less than 80 minutes
With Xpert® MTB/RIF Ultra as frontline diagnosis we are moving a step closer towards TB elimination. It’s a game changer for overcoming the challenge of fast diagnosis of TB in smear-negative patients.
Daniela Maria Cirillo, M.D., Ph.D.
WHO collaborating Centre and TB Supranational Reference Laboratory
San Raffaele Scientific Insitute, Italy
The Need
In 2015, 10.4 million people developed active tuberculosis (TB) (11% of which were HIV positive) and 1.4 million people lost their lives to the disease.1
Today, the challenge to the global TB community is to ‘End TB’. The reliance on microscopy, with a sensitivity of only 46%–78%,2 is a severe limitation. Between 36%–59% of pulmonary TB cases are smear-negative/culture-positive and the relative transmission rate (10-24%) of these patients has a strong impact on patient management in hospitals, as well as TB control programs in developed and developing countries.3,4
In order to support the goal of TB elimination, we must implement efficient active case finding. A precise and early detection of TB is needed to improve case management and significantly enhance prevention of TB transmission.
The Solution
Enhancing the Standard
Xpert® MTB/RIF revolutionized the management of Mycobacterium tuberculosis (MTB) infections by providing faster and more accurate MTB diagnosis that detects MTB and rifampicin (RIF) resistance simultaneously. Following World Health Organization endorsement in 2010, Xpert MTB/RIF
has helped improve TB programs in over 130 countries and counting.
Building on this success, faster and more accurate detection of MTB from the first point of encounter in the community is critical. Partnered with GeneXpert® Systems, Xpert MTB/RIF Ultra will bring:
- Improved performance and faster time to result
- Higher sensitivity especially in smear-negative TB cases
- Results in <80 minutes5
- Increased accuracy of Rifampicin results
- Improved detection of mixed infections
- Same easy-to-use process
The Impact
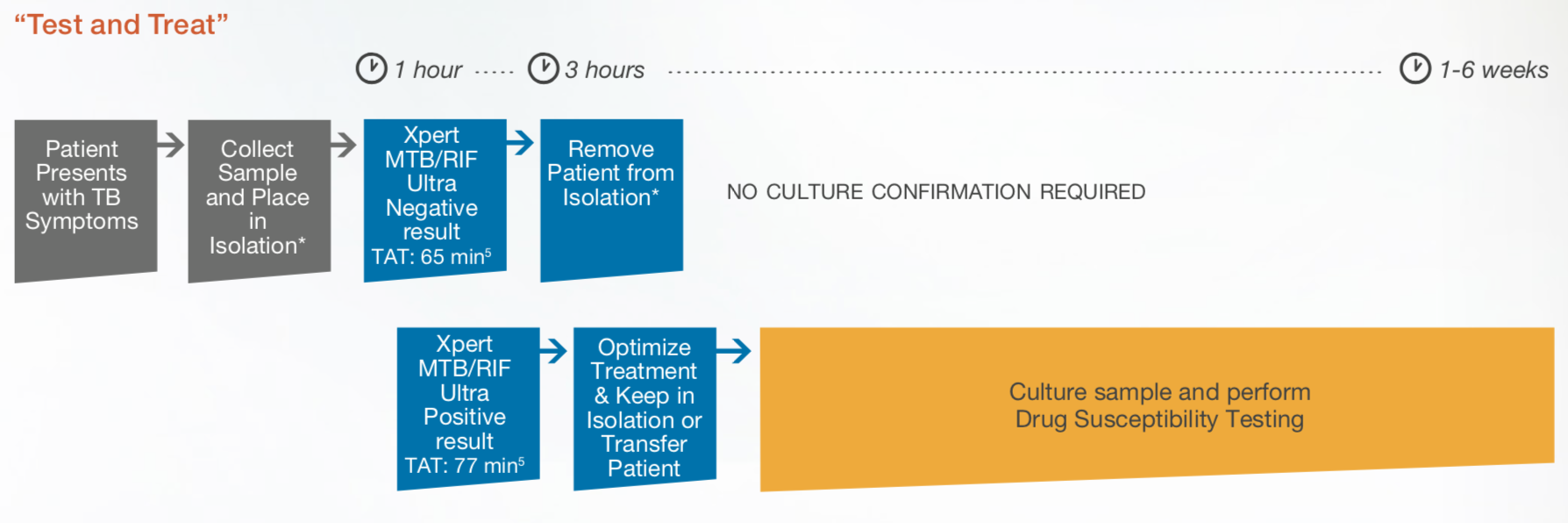
Preventing the transmission of MTB by fast identification of contagious patients, placing them in airborne infection isolation and initiating the right antibiotic therapy as soon as possible are crucial to ‘END TB’. Recently published recommendations by global experts demonstrate the efficiency of using Xpert® MTB/RIF Ultra as a frontline test over smear microscopy or line probe assays followed by culture-based methods for drug susceptibility testing.7,8,9 Thus, a single modification of the diagnostic algorithm to detect smear-positive and smear-negative TB patients improves active case management, is more cost-efficient3 and could potentially reduce the relative rate of transmission. Using Xpert MTB/RIF Ultra has the potential to reduce the number of missed smear-negative TB patients and may lead to a decrease in total costs of patient care due to the detection of MTB in patient specimens and RIF-resistance in a single test.
Impact on Patient Management Pathway
Clinical Impact
- Early identification of TB in suspected patients
- Faster detection of drug-sensitive and -resistant TB cases greatly improves initiation of appropriate treatment
- Cost-efficient case management3
- Enhanced performance in HIV-positive patients and children for a fast active case finding10
- Improved patient outcome
Laboratory Impact
- Faster time to result
- Lower number of culture confirmations due to the high negative predictive value of Xpert MTB/RIF Ultra
- Enhanced sensitivity over smear microscopy of MTB-complex in pulmonary samples
- On-site and on-demand test availability
- Reduced complexity and labor requirements
View Brochure View Safety Data Sheet View Full Details
References
- World Health Organization. Global Tuberculosis Report 2016. http://apps.who.int/iris/bitstream/10665/250441/1/9789241565394-eng.pdf?ua=1
- Kox. Tests for detection and identification of mycobacteria. How should they be used? RespirMed.1995Jul;89(6):399-408.
- Dieletal.Cost-benefit analysis of XpertMTB/RIF for tuberculosis suspects in German hospitals.EurRespirJ.2016Feb;47(2):575-87.
- Behretal.Transmission of Mycobacterium tuberculosis from patients smear-negative for acid-fast bacilli.Lancet.1999Feb6;353(9151):444-9.
- Jones M, et al. Xpert® MTB/RIF Ultra - design and analytical performance of a second generation Xpert® MTB/RIF assay. Poster presented at ECCMID. 2016 April 9-12,
Amsterdam, Netherlands - Uplekaretal. WHO’s new End TB strategy. Lancet.2015May2;385(9979):1799-801.
- Global Laboratory Initiative.GLI model TB diagnostic algorithms. Accessed Mar2017.http://www.stoptb.org/wg/gli/assets/documents/GLI_algorithms.pdf
- World Health Organization. European Tuberculosis Laboratory Initiative. Algorithm for laboratory diagnosis and treatment-monitoring of pulmonary tuberculosis and drug-resistant tuberculosis using state-of-the-art rapid molecular diagnostic technologies. Accessed Mar 2017. http://www.euro.who.int/__data/assets/pdf_file/0006/333960/ELI-Algorithm.pdf
- Global Laboratory Initiative. Planning for country transition to Xpert®MTB/RIFUltra cartridges. AccessedApr2017.http://www.stoptb.org/wg/gli/assets/documents/GLI_ultra.pdf
- World Health Organization. WHO Meeting Report of a Technical Expert Consultation: Non-inferiority analysis of Xpert MTB/RIF Ultra compared to Xpert MTB/RIF. Accessed Mar 2017.
http://apps.who.int/iris/bitstream/10665/254792/1/WHO-HTM-TB-2017.04-eng.pdf?ua=1